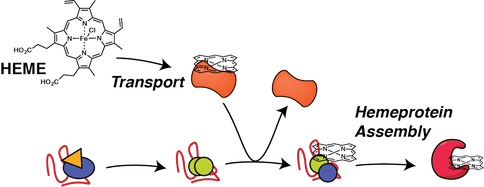
Hemeprotein proteostasis and dysregulation by stress and disease
Biogenesis of cofactor containing proteins requires careful timing of cofactor insertion and coordination with proteostasis network components that aid in the protein folding process. Heme is one of the most common and most versatile cofactors, but free heme is highly toxic due to generation of reactive oxygen species. Therefore, heme has to be securely chaperoned and trafficked to the sites of heme protein synthesis in different organelles, yet specific heme transport factors are largely unknown. Our research identifies intracellular heme transport factors and heme chaperones, and defines how these interact and coordinate with the proteostasis network to facilitate timely insertion of heme during protein folding. Investigating how heme transport and and hemeprotein assembly is regulated during stress and disease will identify new targets to mitigate oxidative damage during cardiovascular disease or to intervene with malaria infections.
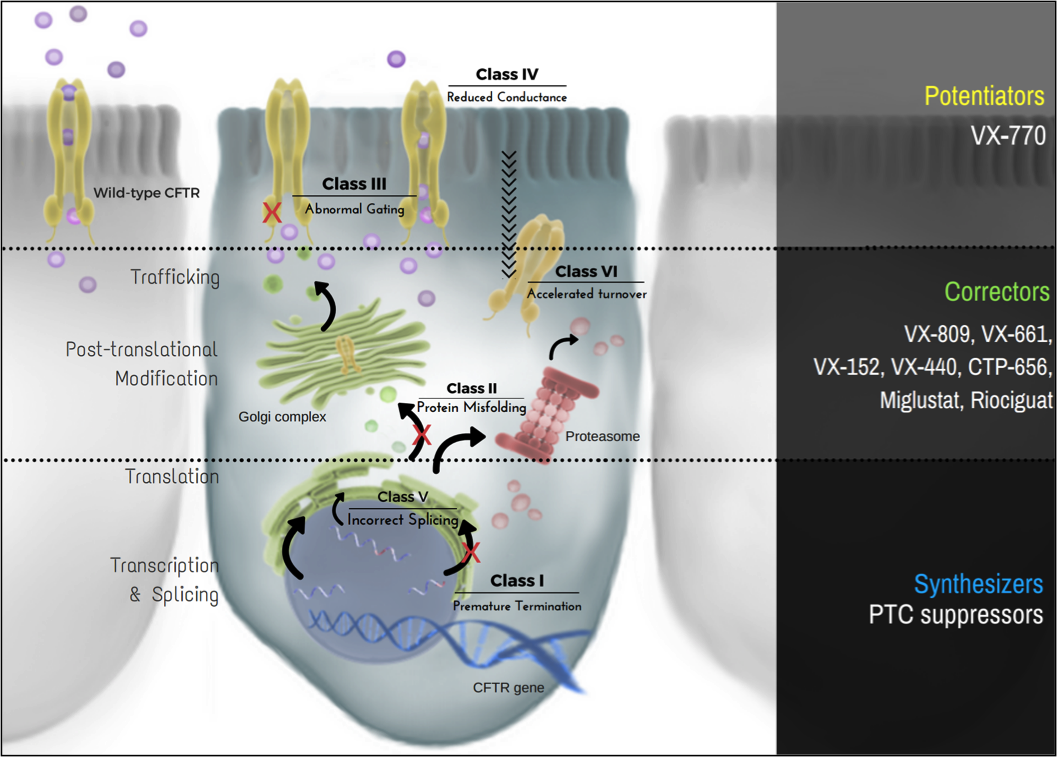
CFTR interactomics to elucidate patient mutation mechanisms and drug response
|
Approved cystic fibrosis corrector drugs improve the folding and trafficking of disease-causing mutant variants of the cystic fibrosis transmembrane conductance regulator (CFTR). These drugs have historically targeted F508del CFTR, the most common disease-causing variant, but they have shown varying efficacy against less common CFTR mutations. The ability to quantitatively compare CFTR interactomes, particularly chaperones and factors involved in protein trafficking, in response to a range of mutations and treatments targeting discrete aspects of CFTR biogenesis will allow us to identify the underlying factors that govern the divergent mutation response to treatment. The specific proteostasis factors/pathways that we identify will be viable targets for future therapeutic intervention directed at targeting the proteostasis network to correct CFTR defects.
|